Updated Coronavirus Boosters Now Available for Children as Young as 5
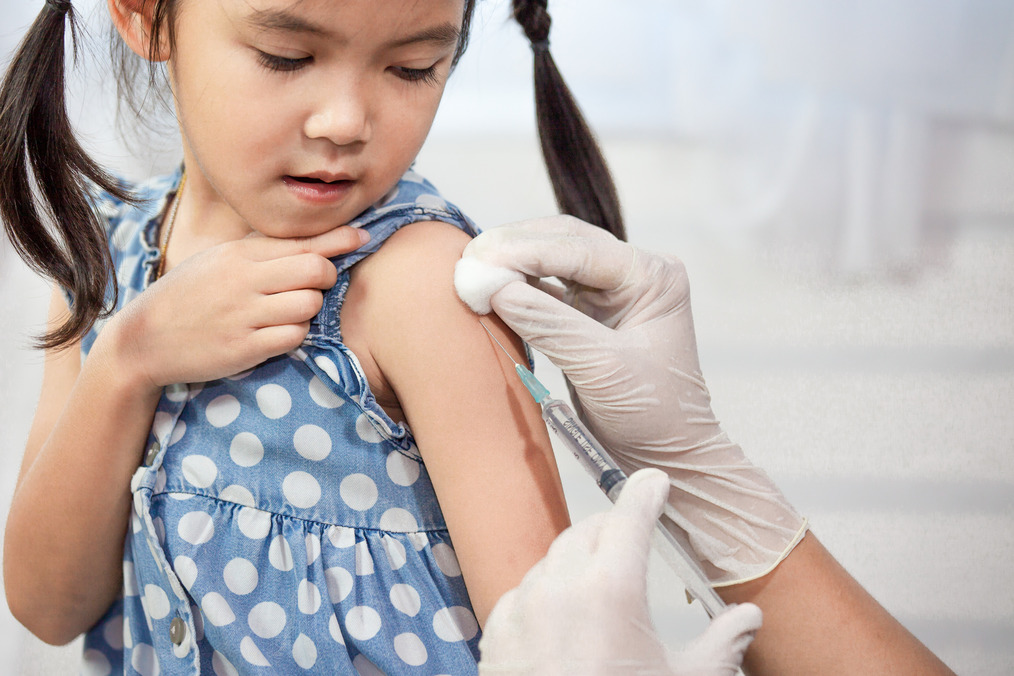
The reformulated boosters are bivalent — they include components of the original strain of the virus and of omicron subvariants BA.4 and BA.5, which make up about 80 percent of the virus circulating in the United States, according to the Centers for Disease Control and Prevention.
The FDA gave emergency use authorization to the Pfizer-BioNTech updated booster shot for children ages 5 to 11. That shot was already cleared for individuals 12 and older. The agency also authorized the updated Moderna booster for children 6 to 17. Previously, it had been authorized for those 18 and older
“Since children have gone back to school in person and people are resuming pre-pandemic behaviors and activities, there is the potential for increased risk of exposure to the virus that causes COVID-19,” Peter Marks, the FDA’s top vaccine official, said in a statement.
Read full article on The Washington Post.




