Chemicals are an important component of laboratory functions. If used carelessly, they can cause severe physical, structural, and/or financial damage to the University and its employees.
These damages may be brought about by an immediate reaction or long-term misuse/neglect of a chemical. To prevent the misuse of chemicals, the employee must identify any hazards associated with the chemical in use. This can be done by reviewing the Safety Data Sheet (SDS) before working with the chemical. Plan work carefully. At the beginning of complex research projects, conduct a risk assessment. Ask these questions:
- What are the hazard(s) associated with the chemicals involved in the research? (Nitric Acid, Xylene, Sodium Azide)
- What kind of PPE is needed? (fume hood, biosafety cabinet, apron, splash-proof goggles)
- Will the process generate waste?
- Will the waste be hazardous? (reactive, ignitable, corrosive, toxic)
- What type of reactions take place when using the chemicals? (generate heat, acid-gas formation, freeze)
Tips for Chemical Storage
Many laboratories find it convenient to store their chemicals alphabetically. This may seem like good lab organization, but it can lead to incompatible hazardous chemicals being stored together. For example, say we store all the chemicals starting with "S" together. What usually happens in this situation is Sulfuric Acid (Strong Acid) gets stored on the same shelf or in the same cabinet as Sodium Nitrite (Strong Oxidizer). If one of these containers leaked (or the shelf falls), an acid gas could result as a reaction between the two chemicals. Consult the chemical's SDS for identifying hazards and compatibility issues. Click here for the Chemical Segregation Tip Sheet.
Storing chemicals by their storage group is a good way to make sure that incompatible chemicals are not stored together. Stanford University created a storage group system that is widely used today to help labs correctly store their chemicals. SciShield's ChemTracker is integrated with the Stanford Storage Group system, so it is easy to see what storage group your chemical belongs to. Once the storage group is identified, use this Chemical Storage Guidelines Chart to see best storage practices:
To find the storage groups on ChemTracker:
1. Log into SciShield and navigate to your lab’s chemical inventory.
2. Click the download button near the top of the chemical inventory. This will export the inventory into an Excel sheet.
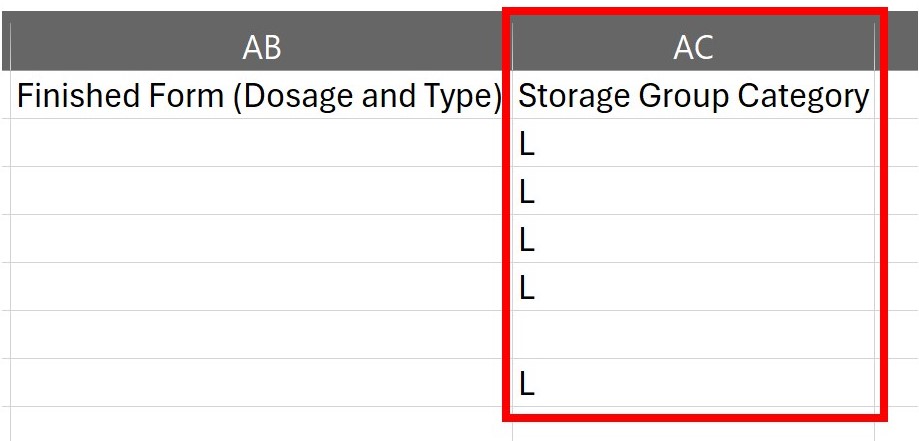
3. In the excel sheet, navigate over to column “AC”. This is where the storage group for each line in the excel sheet is located.
Chemical Segregation Frequently Asked Questions
Q: Why can’t I store my chemicals alphabetically?
A: There can be multiple incompatible chemicals that all start with the same letter. For example, acetic acid and ammonium hydroxide both start with “A” but are incompatible with each other, and should not be stored together.
Q: Why should I check the SDS for storage recommendations?
A: While the Stanford storage guide proves recommendations, there are always exceptions to the rule. EHS can provide more guidance for the storage of materials that may not fit into the defined storage groups.
Q: I don’t see any GHS symbols on my container.
A: Chemicals manufactured before June 2015 are not required to have GHS symbols on the bottle, so do not assume that the chemical is not hazardous. The best way to check a chemical’s GHS symbols is to look at the SDS.
Q: Where can I get the SDS for my chemical?
A: EHS subscribes to the Chemwatch SDS Repository, which can be accessed with your UMID login. You can also look at the chemicals manufacturer’s website for the SDS.
Q: What is secondary containment?
A: Secondary containment refers to a control system that prevents leaking and spills from leaving a specific area. For chemical storage purposes, secondary containment can be a bin that you use to keep incompatible materials from accidentally coming into contact with one another.
Q: What should I use as secondary containment?
A: HDPE is the most chemical resistant plastic and is recommended for secondary containment.
Q: What if I don’t have a flammable or corrosives cabinet?
A: Flammable cabinets are required for more than 10 gallons of flammable liquids. Corrosive cabinets are not required but are recommended. Many fume hoods come with chemical storage cabinets on the bottom.
Q: Where can I find the storage group for my chemicals?
A: ChemTracker is integrated with the Stanford Chemical Storage system. If you use the export function on ChemTracker, it will create a spreadsheet. The very last column of that spreadsheet has the storage code for that chemical. You can use this chart to interpret what the code means.
Q: What if there is no storage group for that chemical?
A: There are a variety of reasons why a storage group might not populate for a particular chemical in your inventory. Please reach out to EHS and we can help troubleshoot the issue and make storage recommendations.
If you require further assistance on how best to segregate your chemicals, contact EHS at 410-706-7055 for a consultation.
What to do with your chemicals if you are moving buildings or leaving the University
If you are leaving the University or moving to another building (or lab space), be sure to remove all unwanted chemicals from the lab before you leave the building (or lab space). The EHS Inspection Team has found chemicals left behind by previous labs (some 20-plus years old) that current users are not aware of, nor have they been trained on their potential hazards. Old or expired chemicals present serious hazards for lab personnel and EHS employees who manage the disposal of these chemicals. Updated chemical inventories and frequent housekeeping will prevent the unnecessary accumulation of chemicals. EHS can provide lab chemical cleanouts if you do not want to take chemicals with you.
Unwanted, Abandoned, or Waste Chemicals
Many of the chemicals used in research laboratories fall under EPA regulations concerning hazardous waste. Once these materials are abandoned, discarded, considered used and no longer wanted, or expired, they should be submitted to EHS for disposal as hazardous waste. Please see Hazardous Material Management for pickup request forms and more information.